Caesium nitride11/6/2023  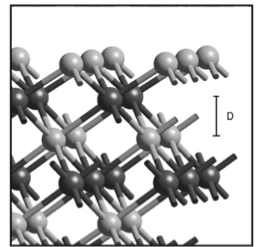
High-pressure nitriding is another method of synthesizing Cs3N, but it usually requires specialized equipment and conditions. To obtain a pure and stable product, the temperature, pressure, and nitrogen flow rate must be carefully controlled. Thermal nitriding of cesium metal in nitrogen gas at high temperatures can synthesize Cs3N. Storage in airtight containers and proper disposal are recommended to minimize potential exposure and hazards. Inhalation or skin exposure to Cs3N dust or fumes can be toxic and cause irritation. It can be hazardous to handle, and appropriate safety measures and precautions should be taken when working with it. Cesium Nitride Safety and HazardsĬs3N is a highly reactive and unstable compound that can decompose at high temperatures. Note: Some of the values are estimates or not experimentally determined due to the instability of Cs3N and the difficulty in handling the compound. The exact details of its structure and properties are still being studied and analyzed by researchers in the field. It is a compound with high thermal stability and potential applications in electronics, optoelectronics, and catalysis. 
It is calculated as the sum of the atomic masses of its constituent elements, cesium and nitrogen.Ĭs3N has a layered crystal structure, with layers of cesium and nitrogen atoms stacked on top of each other. The molecular weight of Cs3N is 412.7 g/mol. However, its density has not been experimentally determined due to its instability and tendency to decompose. The density of Cs3N is estimated to be around 5.2 g/ml. It has a layered crystal structure, and its melting point is dependent on its synthesis conditions and the purity of the compound. The melting point of Cs3N is estimated to be around 1000☌. The boiling point of Cs3N has not been experimentally determined due to its instability and tendency to decompose at high temperatures. It is calculated as the sum of the atomic masses of its constituent elements, cesium (132.905 g/mol) and nitrogen (14.01 g/mol). The chemical formula for Cesium Nitride is Cs3N, which is composed of one Cesium atom and three Nitrogen atoms. IUPAC NameĬesium Nitride Properties Cesium Nitride Formula It has potential applications in fields such as electronics, optoelectronics, and catalysis. Therefore, there is no excess reactant left in this case.Cesium Nitride (Cs3N) is an inorganic compound composed of cesium and nitrogen, with a layered crystal structure and high thermal stability. The negative value indicates that there is no cesium left over after the reaction and that all of it was consumed. The amount of cesium remaining after the reaction is: The amount of cesium used up in the reaction is: Since we have more moles of N2 than required for the reaction, nitrogen is in excess and cesium is the limiting reactant. 
Moles of N2 = 50 g / 28.0 g/mol = 1.786 molesĪccording to the stoichiometry of the balanced equation, 1 mole of N2 reacts with 3 moles of Cs to produce 1 mole of Cs3N. 
To determine the limiting reactant and the excess reactant, we need to calculate the number of moles of each reactant present: The stoichiometric ratio of cesium to nitrogen in the reaction is 3:1, meaning that 3 moles of cesium reacts with 1 mole of nitrogen to form 1 mole of cesium nitride. The balanced chemical equation for the synthesis of cesium nitride, Cs3N, from cesium and nitrogen is: 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
